 Gabor (1946) was probably the first to realize that the Uncertainty Principle translates into information theory and signal processing. This troubling insight is the heart of the so-called Copenhagen Interpretation of quantum theory, which Einstein was so famously upset by (and wrong about). The Uncertainty Principle does not limit what we can know, it describes the way things actually are: an electron does not possess arbitrarily precise position and momentum simultaneously. 
The most important thing to understand about the Uncertainty Principle is that, while it was originally expressed in terms of observation and measurement, it is not a consequence of any limitations of our measuring equipment or the mathematics we use to describe our results. At the instant at which the position of the electron is known, its momentum therefore can be known only up to magnitudes which correspond to that discontinuous change thus, the more precisely the position is determined, the less precisely the momentum is known, and conversely. 
This change is the greater the smaller the wavelength of the light employed, i.e., the more exact the determination of the position. Here’s how Werner himself explained it:Īt the instant of time when the position is determined, that is, at the instant when the photon is scattered by the electron, the electron undergoes a discontinuous change in momentum. Heisenberg’s Uncertainty Principle is one of the cornerstones of quantum theory. In this article, I look at what exactly we mean by 'resolution' in this context, what the limits of resolution are, and what on earth quantum theory has to do with it. Perhaps surprisingly, Heisenberg’s Uncertainty Principle is sometimes cited as a basis for one technique having better resolution than another. 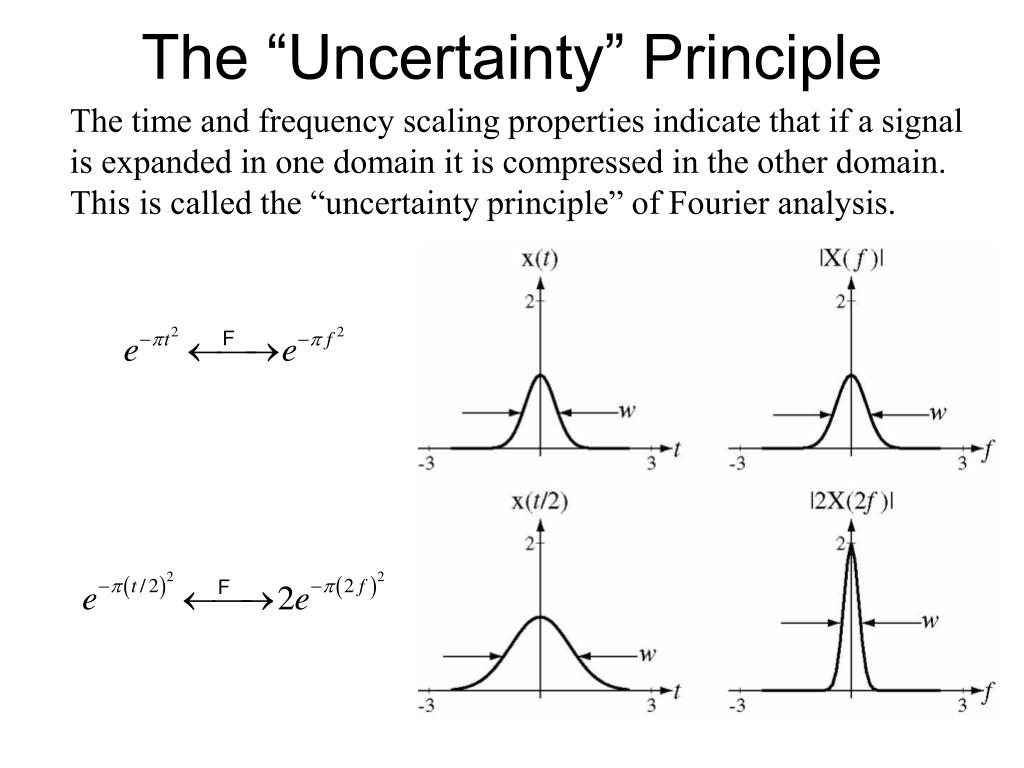
There is frequent mention of the ' resolution' of these various techniques. To date, most of this work has been done with the windowed (‘short-time’) Fourier transform, but other ways of computing spectra are joining our collective toolbox: the S transform, wavelet transforms, and matching pursuit decomposition (e.g. It has also been applied to direct hydrocarbon detection (Castagna et al. Most often applied qualitatively, for seismic geomorphologic analysis (eg Marfurt & Kirlin 2001 ), it is increasingly being applied quantitatively, to compute stratal thickness (Partyka et al. Also time and energy form a Fourier transform pair, so also they obey the same law.Over the last five years or so, spectral decomposition has become a mainstream seismic interpretation process. In particular, we can never know both of them simultaneously with arbitrary accuracy. 
Then the expectation of the placement of the particle is $x_0:=\int_$ in physical units, where $h$ is Planck's constant. The function $|\psi|^2$ is the probability density function of the particle, that is, the probability of finding the particle in a set $A$ is $\int_A|\psi(x)|^2 dx$ and $\psi$ is normalized to have $L^2$-norm equal to $1$. The quantum mechanical situation is the following: A particle in space is represented by its wave function $\psi$, which is complex-valued (and satisfies Schrödinger's equation). One should of course mention a few words about the quantum mechanical interpretation of the Heisenberg uncertainty principle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
